Zinc(II)-Terpyridine based Fluorescent Probes for Selective Detection of Pyrophosphate in Aqueous Medium and Living Cells
Main Article Content
Article Sidebar
Abstract
The selective recognition and precise detection of pyrophosphate (PPi) anion by artificial chemosensors remain an attractive and significant area in analytical science as well as supramolecular chemistry owing to their key role in many cellular and metabolic processes [1, 2]. The abnormal PPi concentration in living organisms leads to several disorders like calcium pyrophosphate dihydrate (CPPD) crystal deposition, chondrocalcinosis and arthritis [3, 4]. Therefore, the development of receptors to bind selectively to PPi ions in physiological conditions and the intracellular PPi concentration in living cells monitoring are highly needed and have gained more attention in recent years [5]. The quantitative determination of PPi using fluorescent chemosensors in water medium is highly selective and sensitive compared to an organic or semi-aqueous medium [6]. However, in the early stages of their development, fluorescent probes are limited to perform sensing of anions in the pure aqueous medium due to their poor water solubility of the receptor and especially PPi detection in water is highly challenging because of its strong hydration energies compared to cations detection. Hence, the design and development of efficient fluorescent probes for the detection of PPi in a 100% aqueous medium is an ongoing challenge. Zn(II) metal ion is non-precious and commonly present in living organisms and Zn(II) based fluorescent probes could be a potential candidate for nucleus staining. The recognition of PPi in water with Di-Zn(II) binding moiety is a particularly selective and sensitive probe for PPi over other nucleotides (AMP, ADP and ATP) than Mono-Zn(II) complex. Furthermore, the symmetric Zn(II) dipicolylamine complexes act as efficient recognition sites for PPi anion and phosphate derivatives due to their strong Lewis acidity and flexible coordination number often overcoming the strong hydration effects of these ions. In addition, the ionic liquids (ILs) have been found to exhibit high fluorescence efficiency with hydrophilic nature and their tuneable property makes them a promising water-soluble fluorescent probe and is expected to be a sensitive and selective sensor for the detection of PPi at lower-level concentrations in aqueous medium [7]. The cationic part of ILs acts as a fluorophore and counter anion would facilitate high hydrophilicity for the fluorimetric detection of PPi in 100% water. Encouraged by these attractive merits, in this work, we report an interesting category of cost-effective water soluble fluorescent probes viz. terpyridine-derived dipicolylamine Di-Zn(II) complexes (TPD-Zn) and ionic liquid-tagged terpyridine based dipicolylamine Di-Zn(II) complexes (TPT-Zn) for the selective and sensitive detection of pyrophosphate (PPi) ions in aqueous environment as well as living cells. The selectivity investigations of both the fluorescent probes towards different nucleotides and anions in HEPES buffer (pH = 7.4) showed highly selective binding towards PPi ions even in the presence of other concomitant nucleotides including ATP, AMP, GTP, CTP and TTP. The emission of TPD-Zn/TPT-Zn can be effectively enhanced by PPi ion via photoinduced electron transfer (PET) inhibition process (Fig. 1A and 1B). The proposed binding mechanism of chemosensors was demonstrated using fluorescence lifetime and 1H NMR spectroscopic studies and also the interaction of TPD-Zn/TPD-Zn with PPi ions were determined by density functional theory (DFT) studies. The probes TPD-Zn/TPT-Zn show an excellent binding affinity towards PPi ions with the binding constants of 7.72 × 105 M-2 and 6.74 × 104 M-2, respectively. Both the probes exhibited a detection limit as low as 0.47 nM and 0.17 nM, respectively. The Job’s plot have shown binding of complexes between TPD-Zn/TPT-Zn and PPi in 1:2 stoichiometry. Also, the quantum yield values and fluorescence lifetime of TPD-Zn/TPT-Zn were lower than those of PPi complexes, revealing the fluorescence enhancement by PPi ions. The biological applicability of TPD-Zn/TPT-Zn as bio-imaging probes for monitoring the intracellular PPi concentration in breast cancer cells (MCF-7) by confocal microscopic imaging and their cytotoxicity have been probed. We believe that the proposed fluorescent sensors using a fluorescence enhancement response will provide a facile platform for the selective and efficient detection of PPi ions in aqueous medium and living cells.
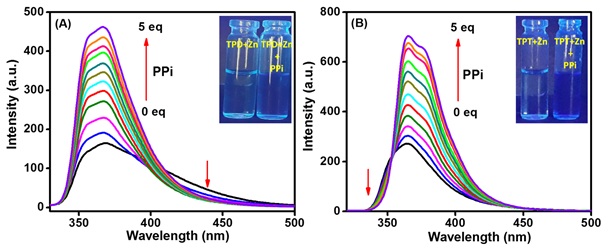
Fig.1. Fluorescence titration spectra of (B) TPD-Zn and (C) TPT-Zn towards PPi (0.5 to 5 eq.) at 1 × 10-6 M concentration in HEPES buffer (pH = 7.4). Inset: Photographs of TPD-Zn/TPT-Zn in the presence and absence of PPi under UV light.
How to Cite
Article Details
Keywords: Ionic liquid, water-soluble, aqueous medium, photoinduced electron transfer, pyrophosphate, living cells.
[2] N. Busschaert, C. Caltagirone, W. Van Rossom, P. A. Gale, Chemical Reviews, 115, 8038-8155 (2015). https://doi.org/10.1021/acs.chemrev.5b00099
[3] A. E. Timms, Y. Zhang, R. G. G. Russell, M. A. Brown, Rheumatology, 41, 725-729 (2002).
https://doi.org/10.1093/rheumatology/41.7.725
[4] J. Ledingham, M. Regan, A. Jones, M. Doherty, Annals of the Rheumatic Diseases, 52, 520-526 (1993). https://doi.org/10.1136/ard.52.7.520
[5] S. Dey, P. K Sukul et al. ACS Omega, 4, 16191-16200 (2019). https://doi.org/10.1021/acsomega.9b02405
[6] D. H. Lee, S. Y. Kim, J. I. Hong, Angewandte Chemie, 116, 4881-4884 (2004). https://doi.org/10.1002/anie.200453914
[7] S. V. Muginova, D. A. Myasnikova, S. G. Kazarian, T. N. Shekhovtsova, Analytical Sciences, 33, 261-274 (2017). https://doi.org/10.2116/analsci.33.261
